Cow manure processed using anaerobic digestion technology can yield biogas rich in methane, which can be used as fuel, a heat source, an electricity generation energy source, or injected into the natural gas grid after purification.
Despite its advantages, the method produces wet residues known as digestate, often utilized as fertilizer but can potentially release greenhouse gases and pollute the air.
To mitigate these environmental threats, a group of UGM faculty members researched a more sustainable digestate management method aligned with the circular bioeconomy concept, capable of generating economic value products beneficial to the environment.
The products include carbon-rich bio-oil, high-value compounds, hydrochar, and liquid phase by-products containing high concentrations of nutrients that can be recycled into fertilizers or soil amendments.
“In addition, the process developed in this research directly processes high-moisture digestate without the need for extensive heat energy-demanding drying processes,” explained Dr. Hanifrahmawan Sudibyo from the Department of Chemical Engineering.
This research was conducted in collaboration with two other faculty members from the Engineering Faculty: Dr. Budhijanto (Department of Chemical Engineering) and Dr. Adhika Widyaparaga (Department of Mechanical and Industrial Engineering).
In addition to internal collaboration, the UGM research team also worked with research partners from Pontificia Universidad Católica de Chile (Chile) and Pontificia Universidad Javeriana (Colombia).
The research findings have been internationally acknowledged through publication in Industrial & Engineering Chemistry Research, a Q1 journal published by the American Chemical Society (ACS) in 2023.
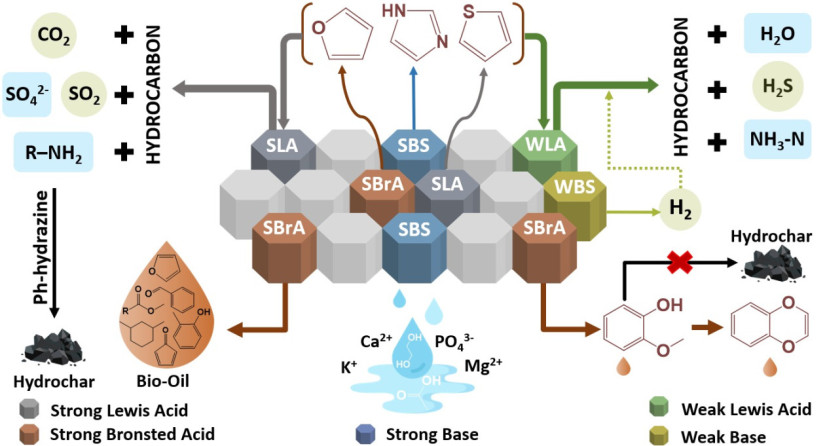
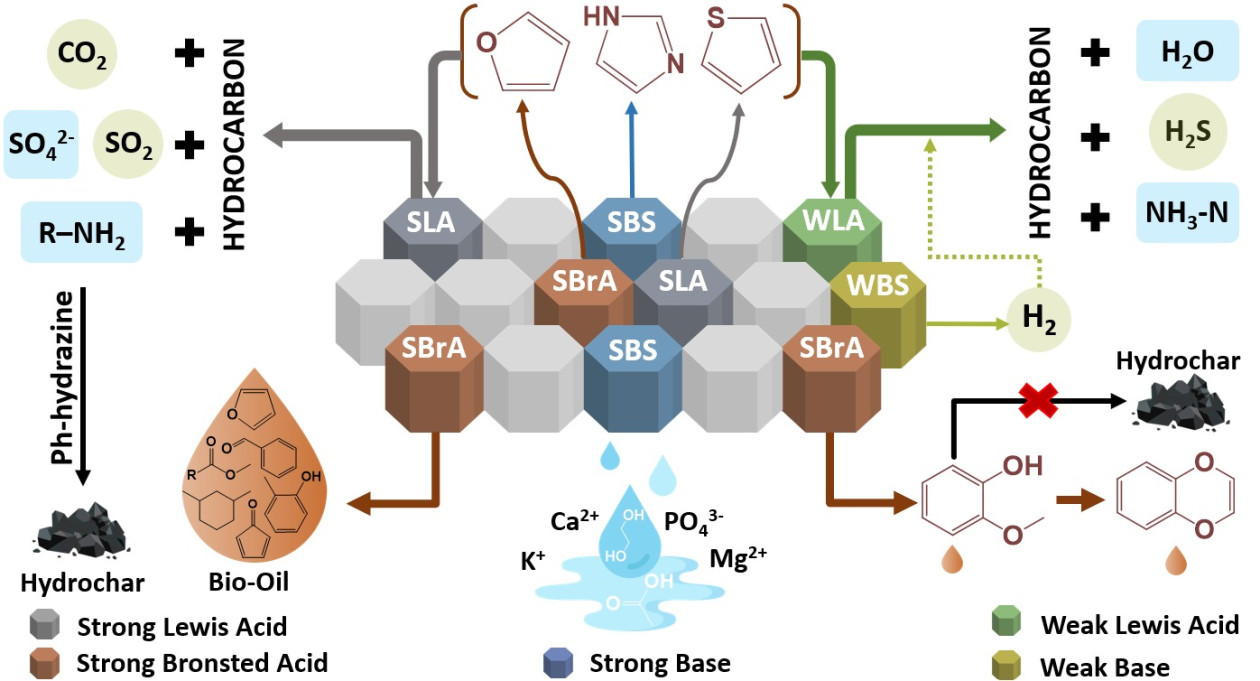
This study used hydrothermal liquefaction (HTL) as the digestate processing method, employing various clay minerals as catalysts.
Dr. Sudibyo explained that the research focused on exploring the most optimal heterogeneous solid catalyst to replace the commonly used homogeneous acid or base catalysts in hydrothermal liquefaction.
The team conducted a comprehensive evaluation of various naturally occurring and commercially available clay minerals and found that montmorillonite was the most suitable for the purpose.
Montmorillonite is a mineral naturally found in various areas across Java, Sumatra, and Nusa Tenggara, among others. World manufacturers have also carried out commercial synthesis of high-purity montmorillonite.
“The results of this research will contribute to developing a resource-efficient biorefinery for processing wet biomass waste, maximizing the recovery of natural resources,” stated Dr. Sudibyo.
The UGM research team plans to investigate further the development of clay mineral-based catalysts for the valorization of various significant wet biomass wastes in Indonesia.
Most recently, Dr. Sudibyo received a research grant from the Kurita Water and Environment Foundation in Japan to develop hydrotalcite-based catalysts impregnated with transition metal elements to convert phenolic compounds in pulp and paper industrial wastewater and leather tanning into syngas, namely CO and H2.
“I hope that the research funded by Kurita will successfully make valuable breakthroughs for relevant industries,” he added.
Author: Gloria